 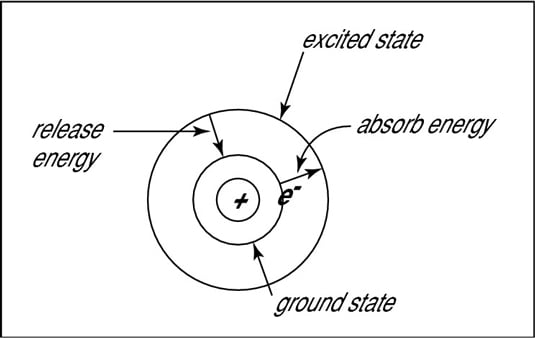
§2 Consider an atom which contains a charge ± Ne at its centre surrounded by a sphere of electrification containing a charge ∓ Ne supposed uniformly distributed throughout a sphere of radius R. A simple calculation shows that the atom must be a seat of an intense electric field in order to produce such a large deflexion at a single encounter…. 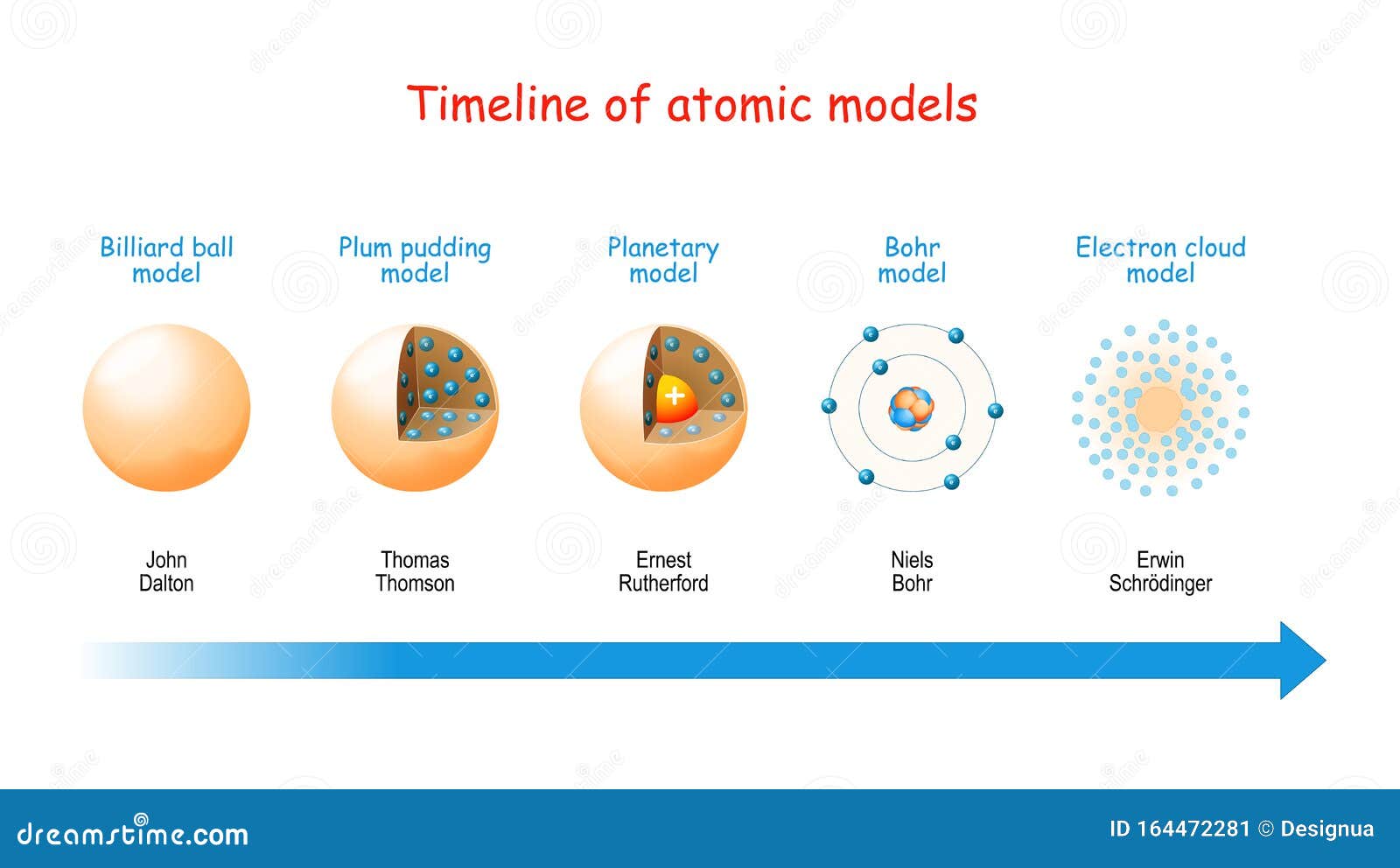
It seems reasonable to suppose that the deflexion through a large angle is due to a single atomic encounter, for the chance of a second encounter of a kind to produce a large deflexion must in most cases be exceedingly small. thick, which was equivalent in stopping-power of the a particle to 1.6 millimetres of air…. §1 The observations, however, of Geiger and Marsden on the scattering of α rays indicate that some of the α particles, about 1 in 20,000 were turned through an average angle of 90 degrees in passing though a layer of gold-foil about 0.00004 cm. There's something positive inside the atomĪ small positively charged region (nucleus)Įlectrons orbit the nucleus like a planet It was left to Rutherford to make conclusions from their observations observation Three different determinations showed that of the incident α-particles about 1 in 8000 was reflected, under the described conditions. To produce a similar effect by a magnetic field, the enormous field of 10 9 absolute units would be required. of gold through an angle of 90°, and even more. If the high velocity and mass of the a-particle be taken into account, it seems surprising that some of the α-particles, as the experiment shows, can be tamed within a layer of 6 × 10 −5 cm. In our experiment, about half of the reflected particles were reflected from a layer equivalent to about 2 mm of air. That sounds like a really difficult task." But then, Geiger and Marsden actually found particles scattered at extreme angles.Įxperiment - alpha particles bombarding gold foil (polonium a source)Ī small fraction of the α-particles falling upon a metal plate have their directions changed to such an extent that they emerge again at the side of incidence.Ĭompared, however, with the thickness of gold which an α-particle can penetrate, the effect is confined to a relatively thin layer. Rutherford's idea was, "well let's see if these guys (Geiger, Marsden etc.) are good at detecting alpha particles scattered from gold foil. Rutherford's assistants did all the work. However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner.Ernest Rutherford (1871–1937) New Zealand–Canada–England It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thus, the Bohr model for heavier atoms described electron shells. Once the level was full, additional electrons would be bumped up to the next level. Bohr believed each electron orbit could only hold a set number of electrons. More electrons were required to cancel out the positive charge of all of these protons. Heavier atoms contain more protons in the nucleus than the hydrogen atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
